 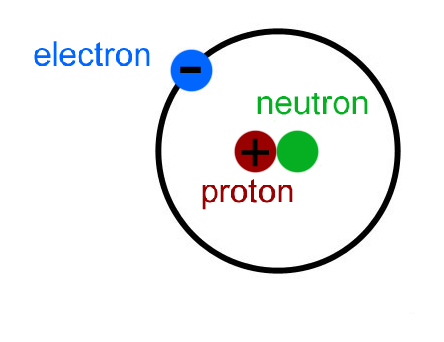 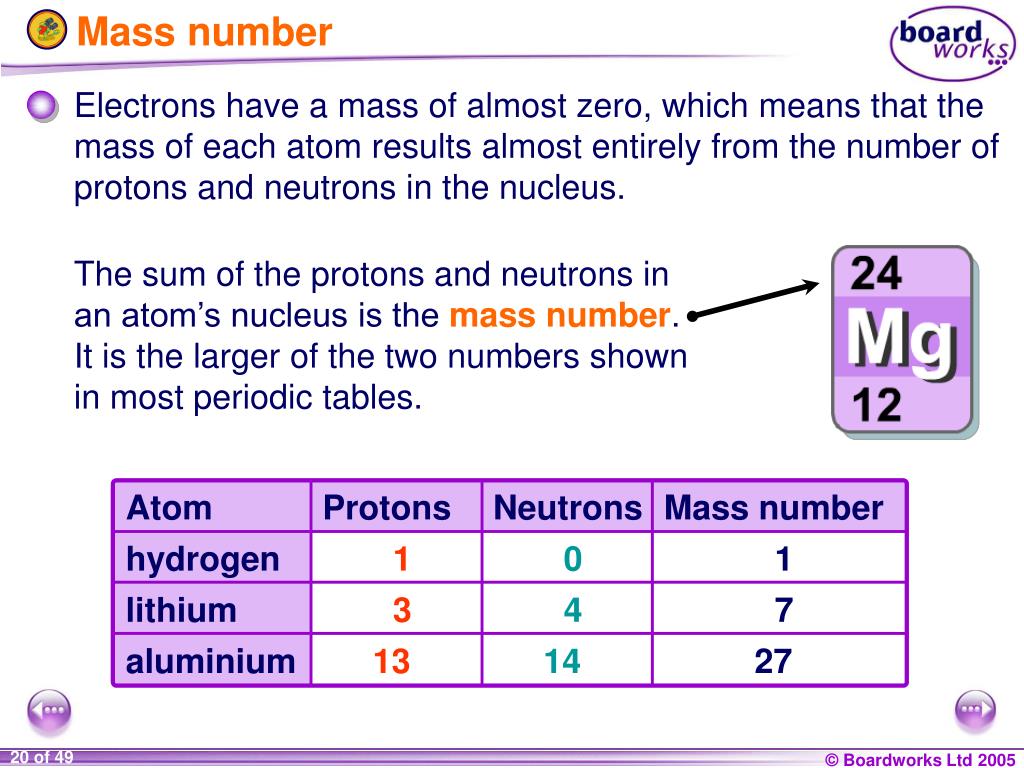 
Much like a proton in mass but without electric charge, the neutron is essential for holding the nucleus together. To avoid this separation, another particle comes into play in the nucleus: the neutron. The same electromagnetic force that draws opposite charged electrons and protons together tries to push the protons (which all have the same charge) away from each other. The number N tells you what element you have: for hydrogen N equals 1, for helium, 2, and so on. The force that holds the electrons and protons together is the electromagnetic force. Basically, it contains a nucleus, holding some number (call it N) of positively charged protons, which is surrounded by a cloud (N) of negatively charged electrons. Swain, professors at the Department of Physics of Northeastern University.įirst it's probably a good idea to review of what an atom is made of. Here, too, neutrons can provide a deep insight.This explanation is provided by the team of Stephen Reucroft and John D. The next step is to study the entire solid with measurements during the charging and discharging process to investigate the interaction of the individual components. Her supervisor at GEMS, Sebastian Busch, is also very satisfied: “The results look very good, and we were also able to see the high mobility of the hydrogen within the solid,” he says. “This measurement at such a precision is not possible with any other method, because neutrons are ideal for studying hydrogen, and additionally they can resolve events in time and thus make them measurable,” explains Neslihan Aslan. For the dynamical processes she turned to the TOFTOF instrument at MLZ for quasielastic neutron scattering (QENS). Neslihan Aslan studied the crystal structure and its stability up to its melting point using synchrotron radiation at the German Electron Synchrotron (DESY) together with Claudio Pistidda from the Institute of Hydrogen Technology. Neutrons reveal high mobility of hydrogen This includes the crystal structure itself, as well as the dynamic processes such as diffusion and rotation of the hydrogen atoms within it. While it is already known that this solid is a good hydrogen storage material, it is not understood why this is so at the microscopic level. Together with colleagues from the Institutes of Hydrogen Technology and Materials Physics at Hereon and MLZ, she has taken a closer look at the compound Li 4(BH 4)(NH 2) 3, which is formed as a product in one of these compounds after hydrogen is released. Neslihan Aslan is a PhD student at the German Engineering Materials Centre (GEMS) of the Helmholtz Centre Hereon at MLZ and conducts research on hydrogen storage systems of this kind. The key is the right mixture of several of these compounds in the hydrogen storage system: By cleverly combining them, it is possible to control via the applied temperature and pressure whether hydrogen is absorbed or released. One type of solid has stood out in particular: compounds of light alkali and alkaline earth metals such as lithium or magnesium, and hydrogen-containing molecules. These solids should be able to store hydrogen in high quantities, and the chemical reactions to produce or release hydrogen should be fast and efficient. It’s the mixture that counts – but what happens microscopically? For this reason, people have long been searching for solids that can chemically bind the hydrogen. This is not practical for many applications, starting with transportation. The problem is that pure hydrogen can only be stored at very high pressure and low temperatures, otherwise it could escape through almost any vessel wall. If one manages to store it, it can be brought to reaction later in order to release the stored energy into electricity. Conversely, it costs energy to produce the hydrogen. The basic principle of these technologies is simple: hydrogen reacts with oxygen to form water, releasing energy in the process. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
